WHY NATIVE COLLAGEN?
HYDROLYSED COLLAGEN
The hydrolysed collagen - component of most cosmetics on the market - is also known as gelatine. It is formed as the consequence of thermal destruction of the cross-linking bonds and peptide bonds of the collagen proteins. The hydrolysate is a mixture of fragments of various weights; however, its molecular mass is always lower than the molecular mass of the native collagen. Because, during the production of gelatine, the delicate structure of collagen does not have to be observed, the raw material for the production of gelatine is subjected to the preliminary processing using strong acids or bases, thus the conditions of production of the hydrolysate are significantly more rapid and enable the use of more raw materials containing collagen, i.e. bones. Because of large amount of cheap raw material, collagen hydrolysate is the cheapest type of collagen derivatives, widely used in the production of foodstuffs as an agent assuring the appropriate texture, or as a thickener. The hydrolysed collagen is also willingly used by the manufacturers of cheaper cosmetics as a substitute to the native collagen protein but it is unfortunately sub-standard.
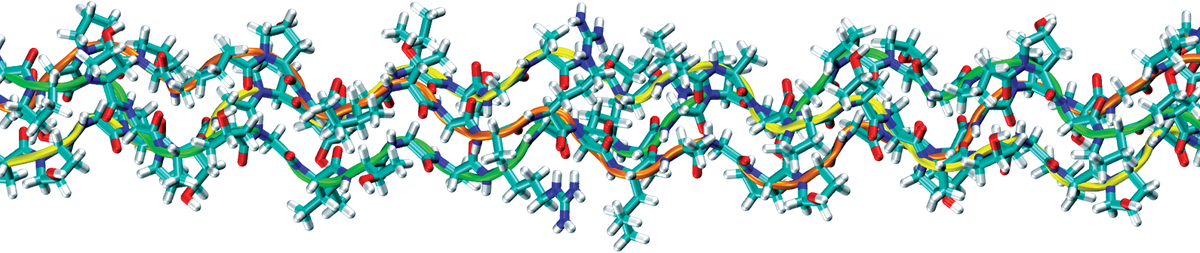
ANIMAL COLLAGEN
Collagen is a group of basic body proteins in animals. The family includes over 20 kinds of proteins of different structures, properties, tissue localisations and functions in the body. These proteins dominate in connective tissue building, e.g. skin, cartilages and bones, and also all important organs. Collagen proteins vary insignificantly among different species; however, the changes have a significant effect on the properties of this protein. Collagen of higher vertebrates is usually more cross-linked and has a higher denaturation temperature. Additionally, during the ageing of collagen of higher animals, the intramolecular and intermolecular lateral covalent cross-linking bonds are formed in their bodies (in vivo) to improve the stability of collagen fibres. The higher the amount of the bonds, the higher the density and resistance of collagen. However, the solubility of collagen is decreased. This collagen cross-linking is disadvantageous for the appearance of our skin. It becomes less elastic and well-toned. The collagen of piscine (fish) hide is significantly less cross-linked than the bovine or porcine hide collagen and thus its solubility is much higher than the others. The higher “solubility” of piscine hides allows for obtaining the native form of collagen in the finished product – collagen gel using an extremely delicate method of obtaining the protein under conservative conditions. The obtaining of collagen from porcine hides requires a very offensive chemical and/or enzymatic processing, causing a significant degradation of the protein. Moreover, the bovine protein may be a prion carrier. Prions are potentially infectious proteins commonly observed in the majority of higher animals (mammals, reptiles and amphibia) that are harmless at the moment of transformation its natural conformation, after which they become proteinaceous infectious particles. The infectious prions cause lethal nerve system diseases in both animals and humans, i.e. BSE or Creutzfeld-Jacob Disease. Fish are lower organisms and until now it was not confirmed that they have the ability to produce prions.
PLANT COLLAGEN (FITOCOLLAGEN)
The plant fitocollagen includes the polycarbohydrates of plant origin giving the skin the effect of a protective film and styptic effect similar to animal collagen. The effect of these compounds is not as complex as the one of collagen. For this case it is just a mental shortcut and an advertising slogan because collagen is a protein and, moreover, a protein produced by animals only. The specific characteristic of collagen is the content of two amino acids – hydroxyproline and hydroxylysine, almost exclusively specific to this protein. However, a plant protein, called extensin, containing hydroxyproline exists. But the structure of this protein is completely different than collagen. Despite the content of the important amino acid – hydroxyproline increasing the moistening of the skin, its effect is similar, not equal, to collagen and thus it may not replace this protein.
MICROCOLLAGEN
The name “microcollagen” is used for usually small proteinaceous particles (oligopeptides) that simulate the fragments of natural collagen. These particles have an effect on the production of collagen and other elements of the dermis. Microcollagen is produced in biotechnological laboratories and modified by the addition of fatty particles that enhance its stability and enable its better permeation through the cutaneous barrier.
LYOPHILISED COLLAGEN - inv native fish collagen
Lyophilisation is one of the methods of obtaining dried products. It is based on freeze-drying and is performed at temperatures below 0°C, under significantly reduced pressure. The process is used for drying highly sensitive compounds and substances, especially those non-resistant to heating. As a result of this process, the active substances keep their activity and valuable elements, i.e. vitamins, proteins, enzymes, minerals, etc., which are not destroyed. The lyophilised preparations contain only trace amounts of water and do not require the use of preservatives. The preparation obtained after lyophilisation may be easily rehydrated without any loss of its organoleptic and biological properties. This type of collagen is used mostly in diet supplements, especially the ones used for regeneration of skin, bones and cartilages. The lyophilised collagen is also used in the production of face-masks and collagen lobes and dressings. All these preparations are usually very expensive, which is the result of high expenditures and the lyophilisation costs.


